Lewy Body Dementia: Symptoms, Diagnosis & Treatments
Key Points
- Lewy body dementia (LBD) is the second most common form of progressive dementia after Alzheimer’s, yet it remains widely underdiagnosed.
- It’s caused by abnormal deposits of a misfolded protein called alpha-synuclein that build up inside brain cells and damage both thinking and movement simultaneously.
- Hallmark symptoms include vivid visual hallucinations, dramatic swings in mental clarity, Parkinson’s-like movement problems, and a sleep disorder where people physically act out their dreams.
- LBD is an umbrella term for two conditions: Dementia with Lewy Bodies (DLB) and Parkinson’s Disease Dementia (PDD) distinguished mainly by which symptoms show up first.
- There is no cure. Treatment is about managing symptoms, and medication decisions are unusually complex many common antipsychotics are dangerous or potentially fatal for these patients.
- Age is the single biggest risk factor, and men are affected slightly more often than women.
What Are Lewy Bodies Dementia?
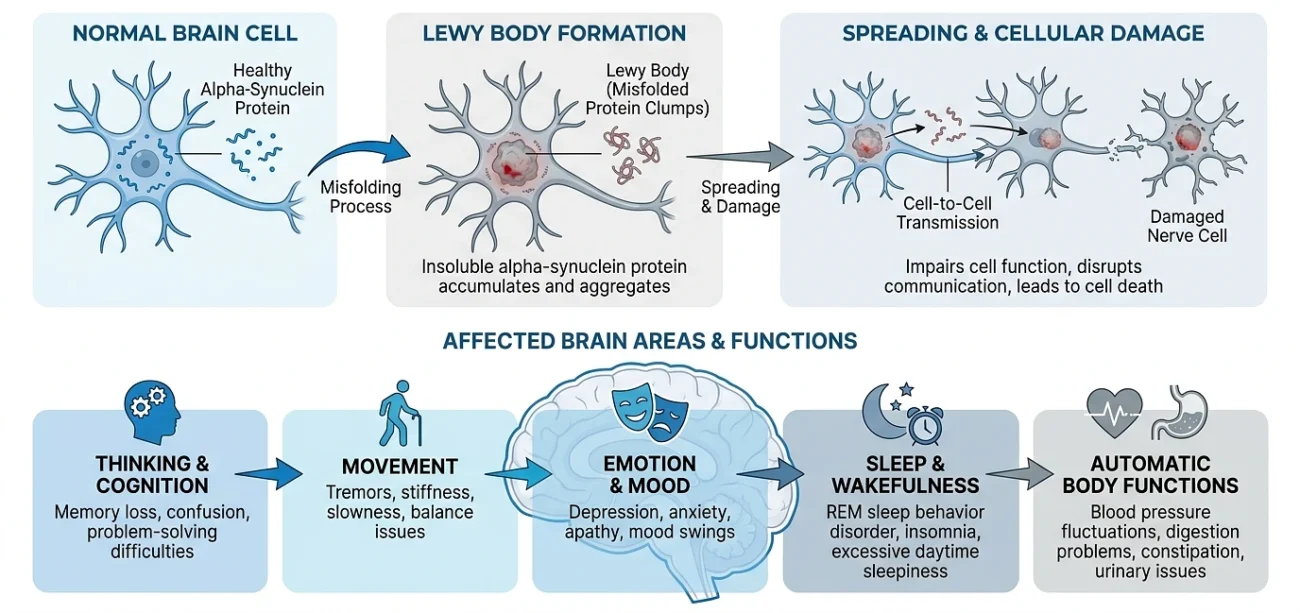
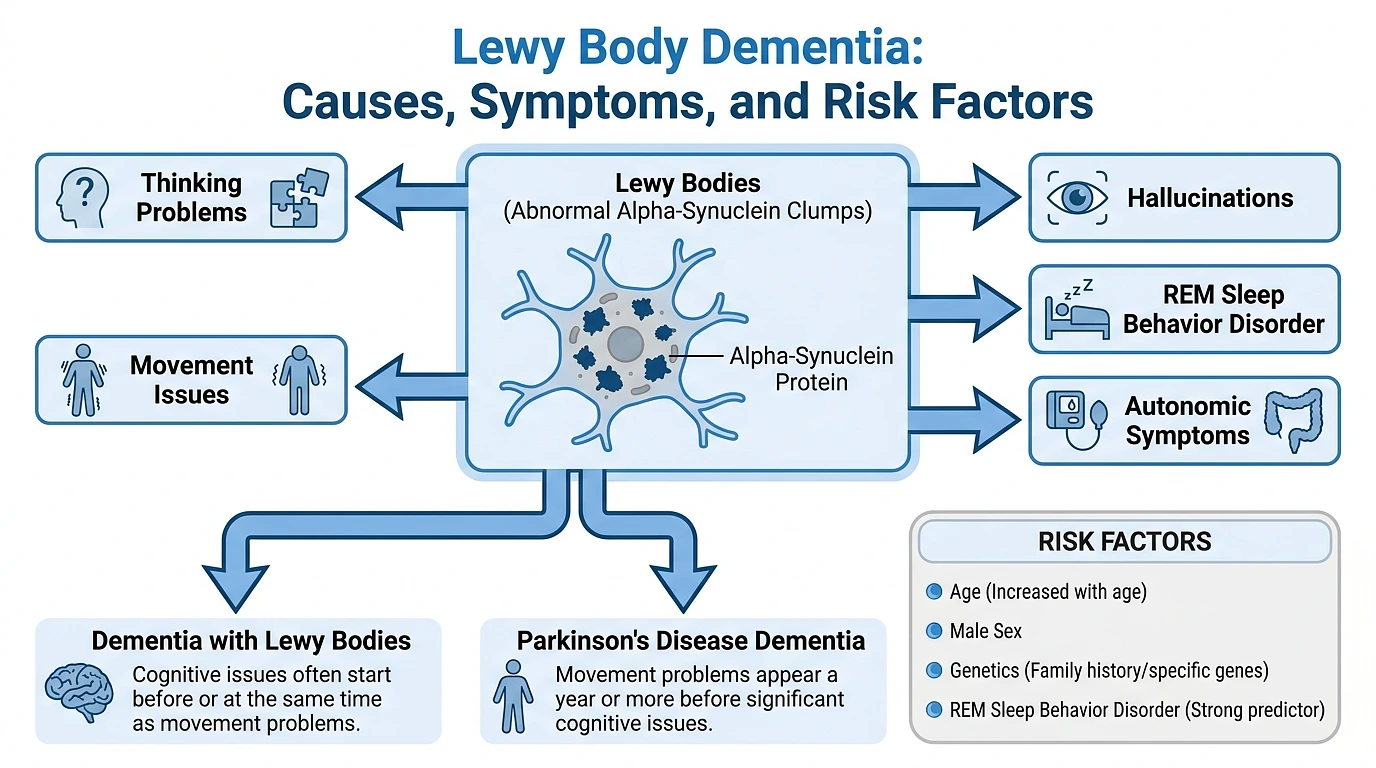
Lewy bodies are tiny, round clumps of a misfolded protein called alpha-synuclein that accumulate inside nerve cells in the brain. In a healthy brain, alpha-synuclein exists in a normal, soluble form and helps neurons communicate with each other.
But when this protein misfolds takes on the wrong three-dimensional shape it starts sticking to itself and forming insoluble clumps. These clumps are the Lewy bodies (Lewy Body Dementia Association, “About LBD: Spectrum, Causes, Risks & Research”).
They were named after Friedrich H. Lewy, a German-American neurologist who first described them in 1912 while working in the laboratory of Alois Alzheimer.
Once formed, Lewy bodies overwhelm the cell’s normal machinery and eventually cause it to die. And they don’t stay in one place. They can form in brain regions that control thinking, movement, emotion, sleep, and even involuntary body functions like blood pressure and digestion (Lewy Body Dementia Association).
That’s why LBD produces such a wide, unpredictable range of symptoms. It’s not just a memory disease. It’s not just a movement disease. It’s both plus a lot more.
There’s also growing evidence that alpha-synuclein spreads between neurons in a prion-like fashion. Misfolded protein in one cell can “seed” misfolding in neighboring cells, gradually expanding the damage across the brain over time (Noguchi-Shinohara M & Ono K, “The Mechanisms of the Roles of α-Synuclein, Amyloid-β, and Tau Protein in the Lewy Body Diseases,” International Journal of Molecular Sciences, 2023).
This may explain why symptoms start small and diversify as the disease progresses the damage is literally spreading.
One more important thing: Lewy bodies are also the hallmark of Parkinson’s disease. The overlap is no coincidence. Parkinson’s and Lewy body dementia exist on the same biological spectrum, sharing the same core protein problem but differing in where the damage begins.
What Causes Lewy Body Dementia & Who Is at Risk?
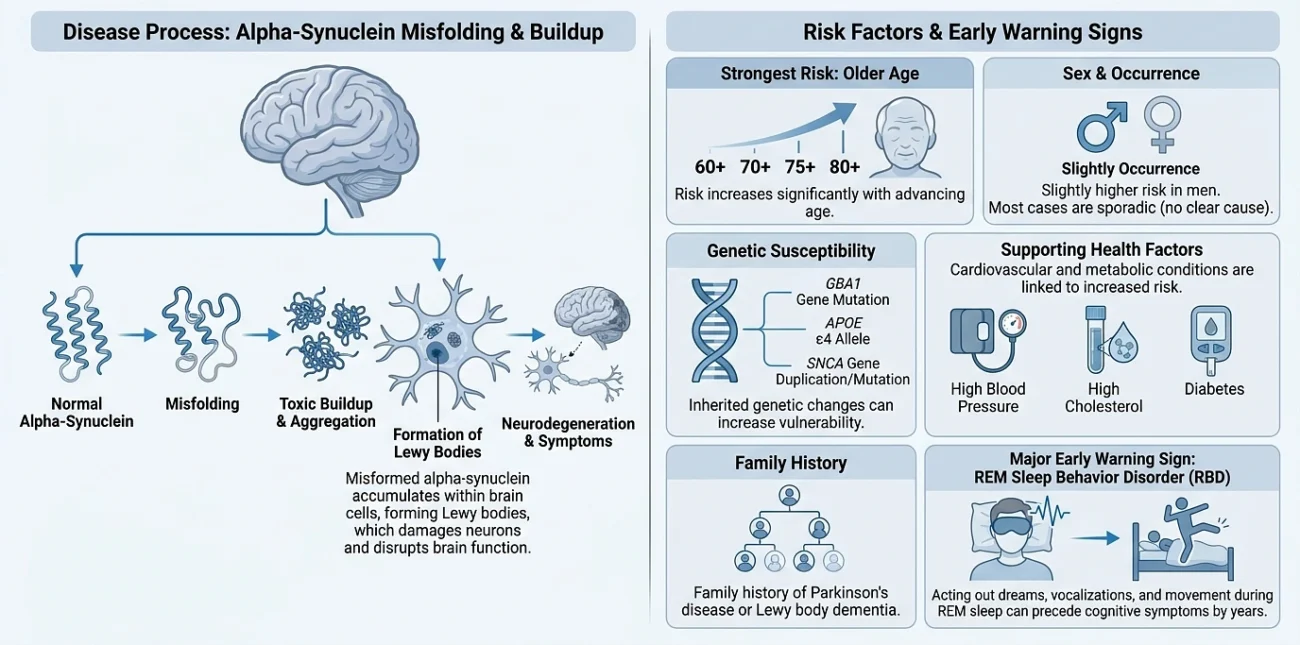
Researchers understand the disease involves alpha-synuclein misfolding and accumulation. But exactly why it happens in some people and not others is still unclear. Almost all cases are “sporadic” they appear without any obvious inherited cause (Alzheimer’s Society UK, “Dementia with Lewy bodies: what is it and what causes it?”).
Age The Biggest Risk Factor
Age is the single strongest predictor. LBD typically develops between ages 50 and 85, with a median onset around age 76. The older you get, the higher the risk (National Institute on Aging, “Lewy Body Dementia: Causes, Symptoms, and Diagnosis”; MedlinePlus Genetics, “Dementia with Lewy bodies”).
People typically survive about 5 to 7 years after diagnosis, though this varies significantly (MedlinePlus Genetics).
Sex
Men are slightly more likely to develop LBD than women. A large systematic review published in 2025, analyzing 167 longitudinal studies, consistently found older age and male sex to be the most commonly reported demographic risk factors for both DLB and Parkinson’s disease dementia (Vik A et al., “Risk factors and predictors for Lewy body dementia: a systematic review,” npj Dementia, 2025).
Genetics Real but Complicated
LBD is not typically considered hereditary. Most people who develop it have no family history. No more than 2% of PD patients, and likely even fewer with DLB, carry a disease-causing mutation in a known gene (Lewy Body Dementia Association).
But certain genetic variants do increase susceptibility:
The GBA1 gene encodes an enzyme involved in the cell’s waste-processing system. Mutations here are among the strongest known genetic risk factors for Lewy body diseases. One European genome-wide study found GBA mutations in approximately 7.8% of DLB cases, with an odds ratio of roughly 8 meaning carriers had about eight times the usual risk (Toft M et al., “GBA and APOE ε4 associate with sporadic dementia with Lewy bodies in European genome wide association study,” Scientific Reports, 2019).
Carriers also tend to have earlier onset, more severe symptoms, and more frequent hallucinations (Coomber E et al., “Exploring the link between GBA1 mutations and Dementia with Lewy bodies,” Neuroscience & Biobehavioral Reviews, 2022).
The APOE ε4 allele long known as the biggest common genetic risk factor for Alzheimer’s also significantly increases DLB risk. It likely contributes to the amyloid-beta pathology found in most DLB brains, though its exact role is still being studied (Alzheimer’s Research UK, “Dementia with Lewy Bodies Risk Factors”; Guerreiro R et al., “Investigation of the genetic aetiology of Lewy body diseases,” Brain Communications, 2024).
The SNCA gene encodes the alpha-synuclein protein itself. Mutations here can occasionally produce a DLB-like picture, but they are rare.
Having any of these risk genes does not guarantee someone will develop LBD. Many carriers remain healthy their entire lives.
Other Risk Factors
High blood pressure, high cholesterol, and diabetes established Alzheimer’s risk factors may also raise DLB risk, though the evidence is less robust (Alzheimer’s Research UK).
Family history of Parkinson’s disease or DLB can increase risk, though most cases are still sporadic (Cleveland Clinic, “Lewy Body Dementia”).
Perhaps the most striking risk marker is a sleep condition called REM sleep behavior disorder. Over 90% of older adults with this condition eventually develop a synucleinopathy DLB, Parkinson’s, or multiple system atrophy often years or decades after the sleep symptoms first appeared (Postuma RB et al., longitudinal iRBD cohort studies). It’s increasingly understood not as a risk factor but as an early sign that alpha-synuclein pathology is already active.
Types of Lewy Body Dementia
“Lewy body dementia” is actually an umbrella term for two closely related conditions. Same underlying pathology. Same protein. Different starting point.
1. Dementia with Lewy Bodies (DLB)
This is the form where cognitive symptoms come first or appear around the same time as movement problems.
A person might start with confused thinking, attention lapses, vivid hallucinations, or unpredictable mental fog before any tremor or stiffness develops. DLB is the more commonly discussed form in clinical settings.
The symptoms that define DLB:
- Fluctuating cognition – Sudden, dramatic shifts in alertness and mental clarity. Sharp and conversational one hour, staring blankly and incoherent the next. These swings can last minutes, hours, or days, and are frequently mistaken for delirium (Johns Hopkins Medicine, “Lewy Body Dementia”).
- Visual hallucinations – Detailed, vivid, and realistic. People often see fully formed images of other people, children, or animals that aren’t there. These aren’t vague shadows they’re specific and recurring. One study found hallucinations in 76% of DLB patients and delusions in 57%, with psychosis frequency climbing from about 50% in mild cases to over 75% in moderate-to-severe cases (Ballard C et al., cited in “The Case for Antipsychotics in Dementia with Lewy Bodies,” Movement Disorders Clinical Practice, 2019).
- REM sleep behavior disorder – The normal muscle paralysis during REM sleep is absent, causing people to physically act out their dreams yelling, punching, kicking, or falling out of bed. Mayo Clinic research found that 75 to 80 percent of men with DLB in their database had this sleep disorder, compared to only 2 to 3 percent of Alzheimer’s patients. It’s one of the strongest markers that distinguish the two diseases (Murray M, Kantarci K et al., research presented at the American Academy of Neurology annual meeting, cited by LBDA).
- Parkinsonism – Muscle rigidity, slow movement, tremor, shuffling gait. Some develop these early. Some late. Some never do.
- Autonomic dysfunction – Blood pressure drops upon standing, chronic constipation, urinary problems, excessive sweating. These happen because Lewy bodies damage the nerves controlling involuntary body functions.
- Severe medication sensitivity – People with DLB can have dangerous, sometimes fatal reactions to common antipsychotic drugs. This is so significant it’s actually listed as a diagnostic feature in the official DLB criteria (McKeith IG et al., Fourth Consensus Report of the DLB Consortium, Neurology, 2017).
2. Parkinson’s Disease Dementia (PDD)
Here, movement symptoms come first. A person gets diagnosed with Parkinson’s disease tremor, rigidity, slowness and then, at least a year later, develops cognitive decline severe enough to qualify as dementia.
This is not uncommon. In one study, nearly 80% of Parkinson’s patients developed some degree of dementia over an 8-year follow-up. Each year, an estimated 14% of PD patients over 65 develop at least mild dementia (Lewy Body Dementia Association, citing longitudinal PD cohort data).
Once dementia develops, PDD and DLB look very similar. The hallucinations, fluctuations, autonomic problems, and sleep disturbances all converge.
How Doctors Tell Them Apart
The rule is simple:
- Cognitive symptoms first (or within a year of movement problems) → DLB
- Cognitive symptoms develop more than a year after Parkinson’s motor symptoms → PDD
(MedlinePlus, National Library of Medicine, “Lewy Body Dementia”)
This is sometimes called the “one-year rule.” Researchers acknowledge it’s somewhat arbitrary both conditions exist on a single biological spectrum of Lewy body disease.
References
- McKeith IG et al., “Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium,” Neurology, 2017.
- Kane JPM et al., “Clinical prevalence of Lewy body dementia,” Alzheimer’s Research & Therapy, 2018.
- Baskys A, “Lewy body dementia: the litmus test for neuroleptic sensitivity and extrapyramidal symptoms,” Journal of Clinical Psychiatry, 2004.
- Ballard C et al., cited in “The Case for Antipsychotics in Dementia with Lewy Bodies,” Movement Disorders Clinical Practice, 2019.
- Vik A et al., “Risk factors and predictors for Lewy body dementia: a systematic review,” npj Dementia, 2025.
- Noguchi-Shinohara M & Ono K, “The Mechanisms of the Roles of α-Synuclein, Amyloid-β, and Tau Protein in the Lewy Body Diseases,” International Journal of Molecular Sciences, 2023.
- Lewy Body Dementia Association, “New Evidence Supports Pimavanserin for Psychosis in Parkinson’s Disease Dementia,” 2024.
- Murray M, Kantarci K et al., REM sleep behavior disorder and DLB research, presented at the American Academy of Neurology annual meeting, cited by LBDA
- MedlinePlus (National Library of Medicine), “Lewy Body Dementia.”
- Alzheimer’s Society UK, “Dementia with Lewy bodies: what is it and what causes it?”
- Cleveland Clinic, “Lewy Body Dementia (LBD): What It Is, Symptoms & Stages.”
- Biggs Institute for Alzheimer’s & Neurodegenerative Diseases, “Lewy Body Dementia.”
